Quantum Mechanics
Molecular quantum mechanics (qm) is used to study the electronic structure of
molecules. Usually I use qm calculations when I don't trust results obtained by
using molecular mechanics (I use qm to verify force field parameters). Other
problems of special interest to medicinal chemists where QM is the preferred
method of choice are protonation state (ph values), tautomers and electrostatic
potential. 
The above equation describes the tautomerisation
between the two indazole tautomers (isomers). There are two nitrogen atoms in
indazole, but only one of them are protonated at physiological ph (7.4, the ph
of human blood ). The below table displays the energy difference between the two
tautomers. Different qm methods and basis sets were used both in gas phase and
aqueous solution. 1H-indazole is found to be the most stable tautomer in
all cases. This is confirmed experimentally by spectroscopic observations in gas
phase. (In this case the wavelengths used in the measurements was probably
verified by a QM calculation)
| qm method |
qm basis |
qm energy kJ/mol |
| DFT(b3lyp)/SOLV |
6-311g**++ |
-8.3 |
| DFT(b3lyp) |
6-311g**++ |
-20.4 |
| HF/SOLV |
6-311g**++ |
-17.3 |
| HF |
6-311g**++ |
-31.2 |
| DFT(b3lyp)/SOLV |
3-21g |
-15.1 |
| DFT(b3lyp) |
3-21g |
-25.6 |
| HF/SOLV |
3-21g |
-27.8 |
HF
|
3-21g |
-41.7
|
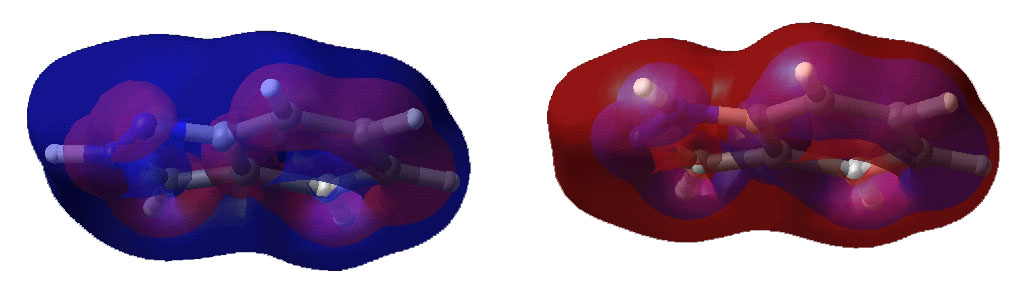
Below are displayed pictures of various surfaces
calculated for the two indazole isomers. These surfaces map different properties
such as electronic potential or total electron density, or they display
molecular orbitals. Comparing property surfaces for different ligands can give
clues to which properties are important for the binding of the ligands to a
protein of interest where as molecular orbitals can reveal details on a
molecules reactivity.
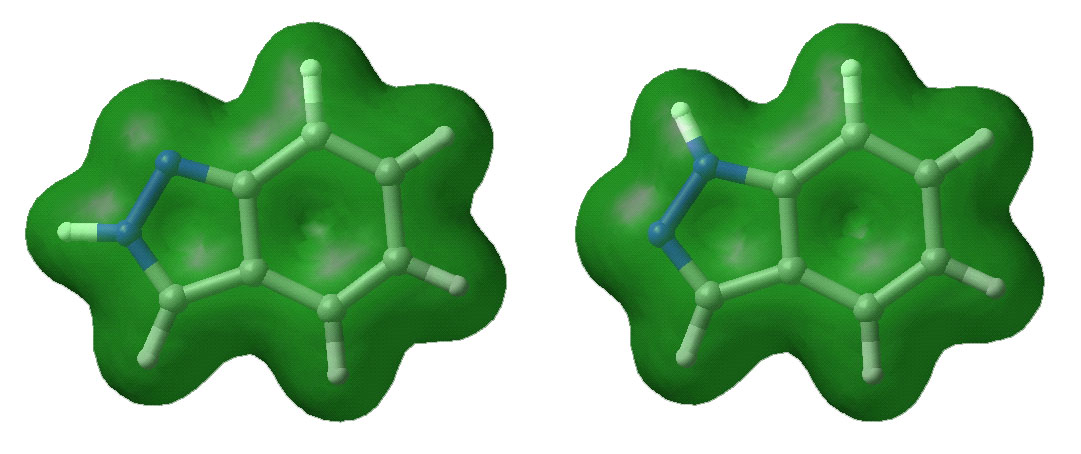
Total electron density surface.
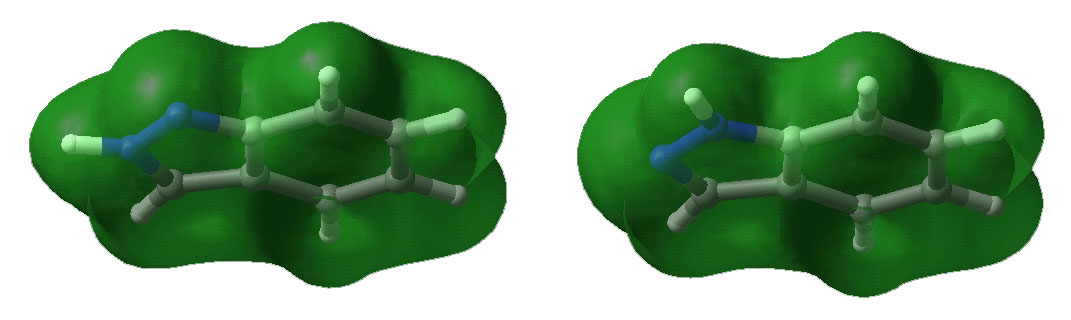
Total electron density surface.
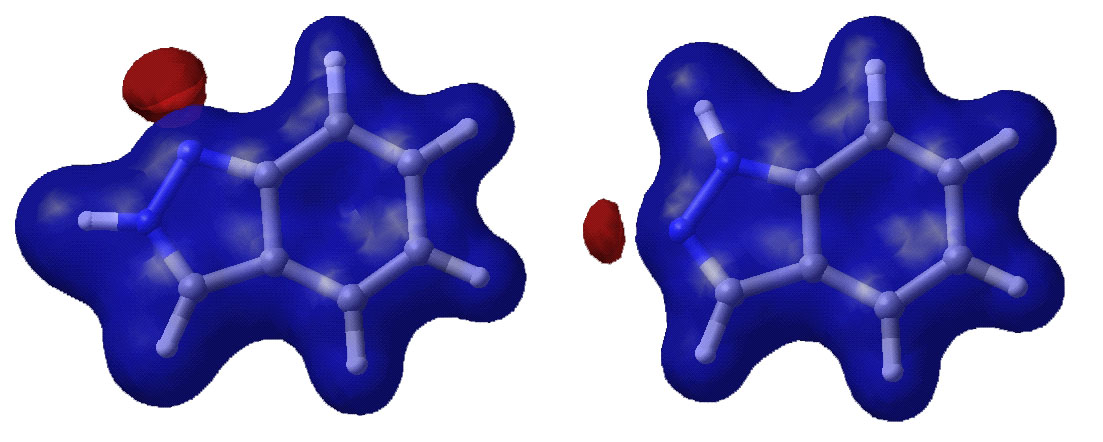
Eectronic potential surface.

Eectronic potential surface.
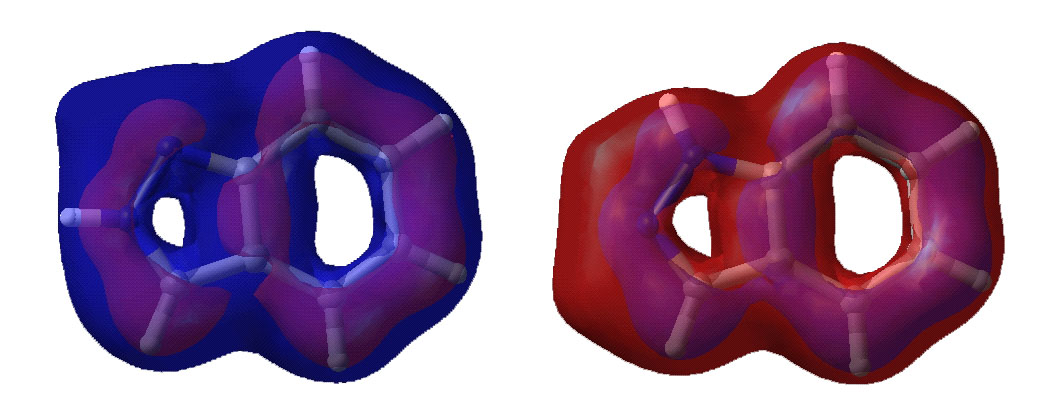
Lowest unoccupied molecular orbital.
Lowest unoccupied molecular orbital.
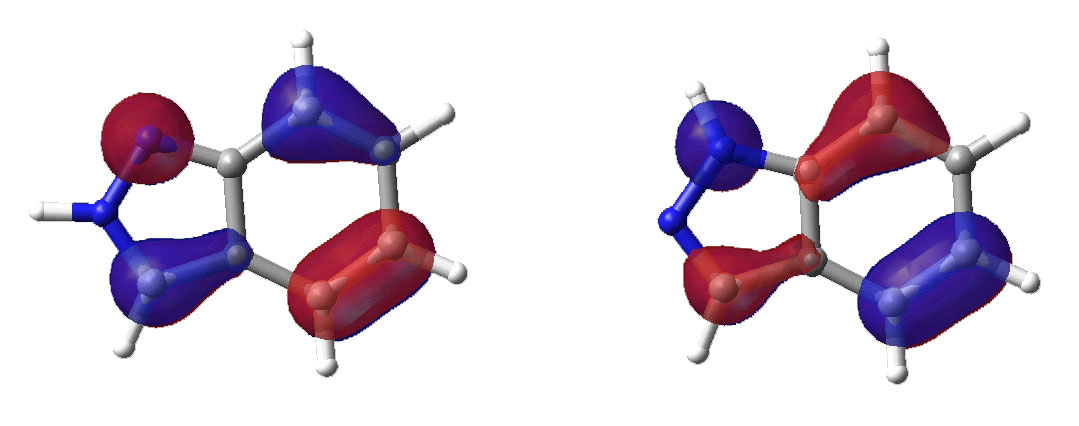
Highest occupied molecular orbital.
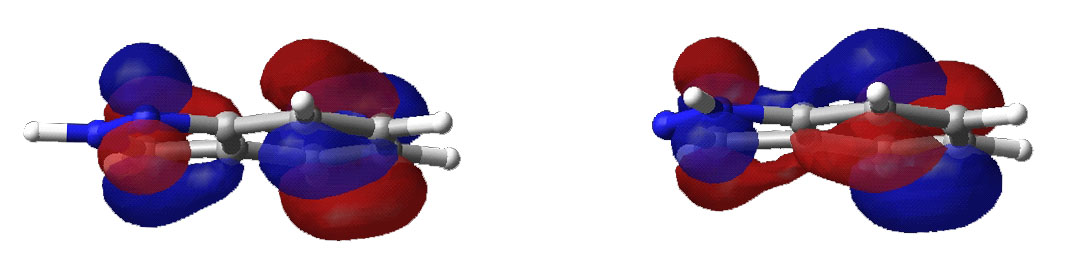
Highest occupied molecular orbital.
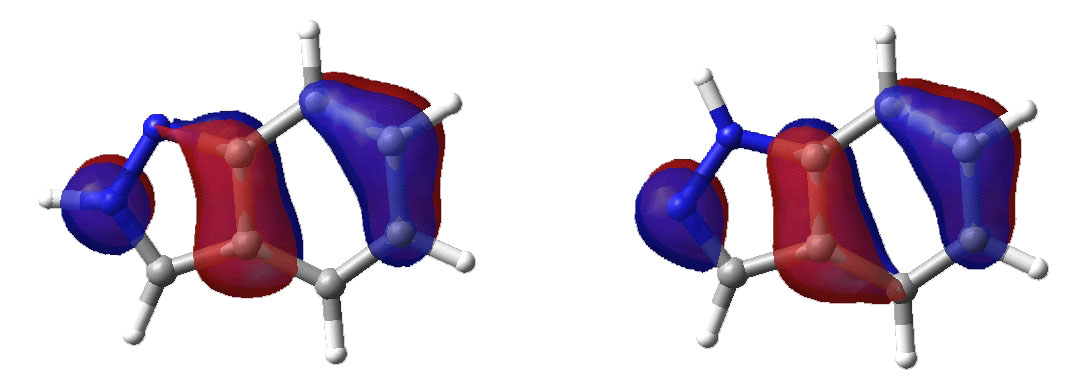
Second highest occupied molecular orbital.
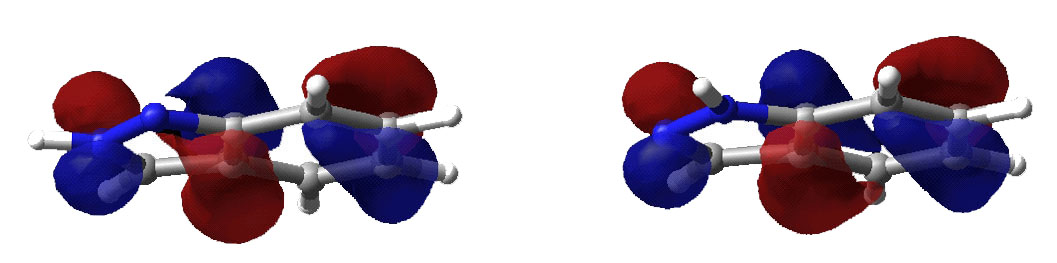
Second highest occupied molecular orbital.
|
